LBCA was excited to learn recently that Dr. Julie Sutcliffe, professor of internal medicine and biomedical engineering at the University of California, Davis had received a $500,000 grant from the 2023 Society of Nuclear Medicine and Molecular Imaging (SNMMI) Mars Shot Research Fund to focus on testing an approach to detect and ultimately treat invasive lobular carcinoma (ILC). We intend to focus on the details of Dr. Sutcliffe’s research project in a future LBCA newsletter but thought it would be useful to speak with her and LBCA Scientific Advisory Board Member Dr. Gary Ulaner in advance of that article to provide information and answer questions about the field of her research: molecular imaging and theranostics and to discuss the promise it holds for ILC treatment. Below we share the questions we asked and their answers.
Q: What is molecular imaging?
According to SNMMI (one of LBCA’s ILC research grant making partners) (see: https://snmmi.org/Patients/Patients/Cancer/Breast-Cancer.aspx), “molecular imaging” is a type of medical imaging that produces pictures of what is happening inside the body on the cellular level, where as other diagnostic imaging procedures such as mammograms, ultrasounds, and MRIs produce pictures of physical structures. Molecular imaging includes the field of nuclear medicine.
Q: What is nuclear medicine?
Nuclear medicine is a medical specialty which uses small amounts of radioactive materials in conjunction with imaging machines to help diagnose and treat disease. Nuclear medicine procedures that individuals with breast cancer may have experienced or heard of include:
- Molecular breast imaging (MBI)
- Sentinel node biopsy (Lymphoscintigraphy)
- FDG-PET (fluorodeoxyglucose-PET)
- FES-PET (fluoroestradiol-PET)
In breast cancer, nuclear medicine has primarily been used for imaging purposes and more recently, with the introduction of FES-PET, to more accurately inform treatment decisions. (To learn more about the FES-PET test and its most appropriate uses click here). In other diseases, nuclear medicine has also been used in the treatment of the diseases.
Q: What is Theranostics? “Theranostics” seems to be a buzzword in oncology these days. Could you break down this term for us? Can you explain how a theranostic intervention works?
Theranostics is a combination of the words therapy and diagnostics. The principle of theranostics is that it starts with performing a diagnostic scan to see if a patients’ tumor expresses a target of interest. And if it does, then you then use the corresponding therapy that has been developed to attack the target (if such therapy exists). In the illustration shown, the beige globe represents the cancer cell and the keyhole depicts the “target” of interest that the tumor is expressing. In this example, research has found a molecule that will “bind” or attach to the target, like a key fitting into a lock. 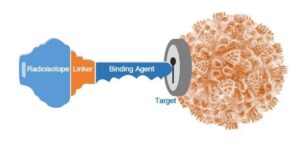
To administer step 1, the diagnostic part of the test, a radioactive molecule is injected intravenously and then the patient is scanned. If this imaging scan is “negative” i.e., the tumor does not light up and can’t be seen, then it means that the cancer tumor is not expressing the target of interest. In such a case, nothing more would be done. However, if the scan results indicate that the tumor does light up meaning the imaging scan is “positive,” the tumor is seen, then step 2 can be taken.
In step 2, the patient will receive a radioactive molecule (also called a radioisotope) intravenously that is different from the one that was used to see if the tumor was lighting up. This radioisotope – the ”therapy” — attaches to the “target” and emits powerful radiation (like a beta or alpha particle) that will kill the cancer cell.
- In this theranostic approach, since the radioisotopes for imaging and the radioisotopes for therapy are injected into the bloodstream, what effect, if any, do they have on the rest of the body? Are the strong radiation-emitting radioisotope treatments only released into the body when they bind with the cancer tumor and do they only go into the tumor, and/or do they go elsewhere?
A: The radioactive molecules travel from the blood to the target of interest. If the target is only present in the tumor then only the tumor will be affected however if the target is also present in other organs then the radioactivity will also go there. It is therefore very important to develop imaging and therapeutic molecules that are VERY specific for the tumor. Radioactivity that is not targeted to the tumor is often excreted through the kidneys so it is very important that patients stay hydrated throughout the treatment to avoid damage to the kidneys.
Q: What are the benefits of treating cancer with a theranostic approach? What are the risks?
With most oncologic therapies, you give a patient a therapy and then wait to see if it will work. With theranostics, you can give the imaging agent first and determine before the patient starts the therapy that if the imaging test is negative, the therapy won’t work. This helps select patients that are likely (or not likely) to benefit from the therapy agent. Following this type of treatment patients can be scanned to see if treatment reached the tumor. This process which uses imaging first helps get the right therapies to the right patients. It also means that the patients who won’t benefit do not waste precious time receiving a treatment that won’t help and can try a different treatment sooner.
Like most therapies there are risks, the main risk associated with these kinds of treatments are due to the radioactivity. Depending on where the radioactivity goes will determine the risks. For example if the radioactivity is excreted or retained by the kidneys there is a small chance that the kidneys will not function as well as before the treatment. There are approaches used to protect the kidneys during the treatment. It is also important that care givers and people in close contact with the patients during the treatment are aware of the risks.
Q: What is on the horizon in the field of breast cancer imaging in terms of molecular imaging?
We currently have FDA-approved ER-targeted molecular imaging with 18F-Fluoroestradiol (FES/PET) for patients with breast cancer. There are several early phase clinical trials evaluating the potential of several different imaging agents targeting different molecules in breast cancer patients including those targeting the molecules: HER2, CD8, SSTR2-, FAP, and αvβ6. Dr Sutcliffe’s SNMMI MARS SHOT grant is evaluating αvβ6-targeted imaging for patients with lobular breast cancer. The αvβ6 is a molecule that is not present on healthy cells but is present on many cancers. Her grant builds on several decades of research by her laboratory developing molecules that can detect and treat cancers sooner. The imaging agent her team has developed, a molecule with the radioisotope gallium-68 attached, has already detected very small lesions including metastasis to brain, bone, liver and lung in patients with pancreatic cancer. In 2023 her team was awarded the Henry N. Wagner, Jr., Image of the Year by SNMMI chosen as an image that best exemplifies the most promising advances in the field of nuclear medicine and molecular imaging. Dr Sutcliffe and her team are now investigating this molecule to image patients with ILC. Their ultimate goal is to identify patients that will be the best candidates for the treatment being developed by her team.
Q: What about anything on the horizon in the field of breast cancer treatment using nuclear medicine approaches?
We do not have any currently FDA-approved radioligand2 therapies for patients with breast cancer. In addition to the several clinical trials evaluating potential molecular imaging targets noted above, there are many clinical trials evaluating the potential of HER2-targeted, SSTR2-targeted, FAP-targeted, and αvβ6-targeted radioligand therapy agents for patients with breast cancer. Dr Ulaner recently published results regarding a successful SSTR2-targeted radioligand therapy (Ulaner et al, Radiology 2024, PMID 39078299). This radioligand therapy has now progressed to a phase II clinical trial called TRACY-1 (NCT06590857). In addition to the ILC focused research already mentioned, Dr Sutcliffe currently has several active clinical trials investigating αvβ6-targeted radioligand therapy in patients with pancreatic cancer (NCT04665947), non-small cell lung cancer (NCT06228482), and most recently a trial investigating radioligand therapy on all metastatic cancers (NCT06389123). The good news is that the landscape is changing rapidly and we can expect new radioligand therapies to emerge.
Citations
1 NCI defines radioisotope as a radioactive isotope, i.e., an unstable form of a chemical that releases radiation as it breaks down and becomes more stable. Radioisotopes can occur in nature or be synthesized.
2 A radioligand is a microscopic particle which consists of a therapeutic radioactive isotope and the cell-targeting compound – the ligand. The ligand is the target binding site, it may be on the surface of the targeted cancer cell for therapeutic purposes.

